 Cryptogamie, Algologie
33 (2) - Pages 105-112
Cryptogamie, Algologie
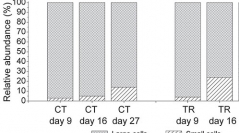
33 (2) - Pages 105-112Bacteria associated to benthic dinoflagellate Ostreopsis cf. ovata cultures were removed to assess their effects on algal growth and toxins' production. Bacteria were removed using an antibiotic cocktail (streptomycin, ciprofloxacin, gentamicin and penicillin G). The actual axenic status of antibiotic treated cultures and bacterial growth in control cultures were assessed by epifluorescence microscopy using SYBR gold dye. The removal of bacteria unaffected algal growth, except conferring a higher cell number at mid stationary phase. Toxin profile and quantification of each toxin (PLTX, OVTX-a, -b, -c, -d, -e) were performed by HR LC-MS on both cell pellet and growth medium extracts. No changes in toxins' profile nor in cell and extra-cellular toxins' concentrations were found between bacteria-free and control cultures at the early stationary phase. Whereas, in late stationary phase axenic cultures showed significant lower cell toxins' concentrations and higher extra-cellular toxins' values, though not significantly (total cell toxins' concentrations: 39.3 and 24.9 pg cell-1; total extra-cellular toxins' concentrations: 23.8 and 28.3 μgL-1 for control and bacteria-free cultures, respectively).